Technical Information on Gold Conjugates
a superior gold probe for light and electron microscopy
| Probe Stability | Long term stability when stored at -20°C or less. (2nm stable for 1 year at 4°C - DO NOT FREEZE) |
| Clustering | 85% singlets with no clusters greater than triplets |
| Concentration and Dilution |
EM probes can be diluted from 1:10 to 1:200. |
| Size Distribution | EM grade probes have a low coefficient of variation, making them quite suitable for multiple labeling applications |
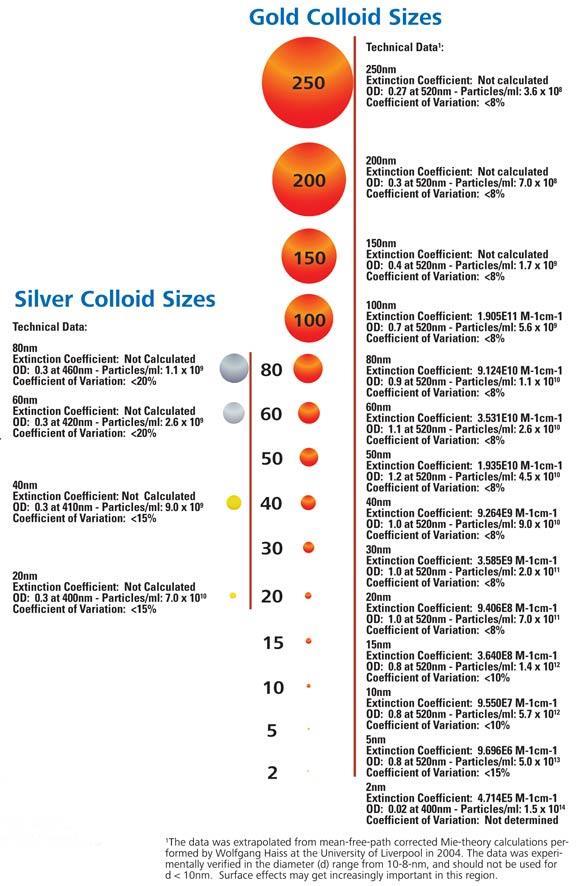
| Particle Sizes | Unconjugated colloidal gold sols come in a wide range of sizes: 2, 5, 10, 15, 20, 30, 40, 50, 60, 80, 100, 150, 200 and 250nm |
Super Ice® Service, when necessary for shipment of heat liable products (e.g. gold conjugates, resins and some fixatives)
Gold particle size will influence labeling density and selection of instrument magnification when used in immunoelectron microscopy. The tables and figures below should aid the researcher in choosing the gold particle best suited to his or her research requirements (reproduced with permission from Giberson and Demaree: The Influence of Immunogold Particle Size on Labeling Density. Microscopy Research and Technique 27:355-357, 1994) .
Table 1: Gold Particle Labeling Densities
| Gold Particle Size | Mean Number Particles/µm2 |
Standard Deviation |
|---|---|---|
| 5nm 10nm 15nm 20nm 30nm |
746 390 227 141 167 |
±105 ±84 ±40 ±26 ±18 |
Table 2: Percent Decrease in Labeling Density for Paired Comparisons of Gold Conjugates
| This Study (1) |
Gu & D'Andrea (2) |
Ghitescu & Bendayan (3#) |
Hansen et al. (4#) |
Henegouwen & Leunissen (5#) |
Yokota (6#) |
|
|---|---|---|---|---|---|---|
| 10 to 5nm | 48% | . | 50% | 46% | 56% | 18% |
| 15 to 5nm | 70% | 73% | 81% | . | . | . |
| 20 to 5nm | 81% | . | . | . | . | 51% |
| 30 to 5nm | 78% | 91% | . | . | . | 80% |
| 15 to 10nm | 42% | . | 62% | . | . | . |
| 20 to 10nm | 64% | . | . | . | . | 40% |
| 30 to 10nm | 57% | . | . | . | . | 75% |
| 20 to 15* | 38% | . | . | . | . | . |
| 30 to 15* | 26% | 33% | . | . | . | . |
| 30 to 20* | none | . | . | . | . | 59% |
| (1) - diameters 5.5, 10.0, 14.6, 20.4, 28.4 (2) - diameters not given, reported as 5, 15, 30 and 40nm (3) - diameters 4.92, 10.1, 15.9nm (4) - diameters not given, reported as 5 and 10nm (5) - diameters 6.7 and 12.1 (6) - diameters not given, reported as 5, 12, 18, 28, and 38nm (#) data extrapolated from graphs and figures to determine percentage decreases * paired comparisons not statistically significant |
Table 3: Paired Comparisons of Gold Particle Size Densities
| Particle Size | . | . | . | . |
| 30 | + | + | 0 | 0 |
| 20 | + | + | 0 | . |
| 15 | + | + | . | . |
| 10 | + | . | . | . |
| .Particle Size | 5 | 10 | 15 | 20 |
| + = Significantly Different at 0.05% Level 0 = Not Significantly Different |
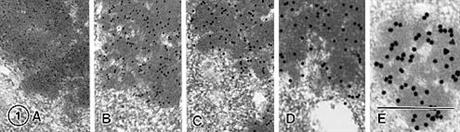
Labeling of the characteristic electron dense granules found in the processed cheese sample is clearly evident for 5 different sized gold conjugates. The magnification for each is identical (60,000).
A: 5nm, B: 10nm, C: 15nm, D: 20nm, E: 30nm, Bar in A-E= 0.5µm
Gold Conjugate Technical Information
| Conjugate | Gold Particle Size |
OD at 520nm | Approximate Protein Concentraion µg/ml |
Approximate Number Gold Particles/ml |
Approximate Proteins Per Gold Particle |
Mean Working Dilution for EM |
| Immunoglobulin | 1nm | - | 50 | 2 x 1015 | 1 to 3 | 1:200 |
| Streptavidin | 1nm | - | 25 | 2 x 1015 | 1 to 2 | 1:200 |
| Immunoglobulin | 5nm | 3.0 | 36 | 1.7 x 1014 | 3 | 1:150 |
| Protein A | 5nm | 3.0 | 12 | 1.7 x 1014 | 4 | 1:150 |
| Streptavidin | 5nm | 3.0 | 20 | 1.7 x 1014 | 5 | 1:150 |
| Poly-L-Lysine | 5nm | 3.0 | 100 | 1.7 x 1014 | unknown | 1:150 |
| Immunoglobulin | 10nm | 3.0 | 30 | 1.7 x 1013 | 12 | 1:100 |
| Protein A | 10nm | 3.0 | 10 | 1.7 x 1013 | 16 | 1:100 |
| Streptavidin | 10nm | 3.0 | 20 | 1.7 x 1013 | 20 | 1:100 |
| Poly-L-Lysine | 10nm | 3.0 | 60 | 1.7 x 1013 | unknown | 1:100 |
| Immunoglobulin | 15nm | 4.0 | 30 | 5 x 1012 | 27 | 1:75 |
| Protein A | 15nm | 4.0 | 10 | 5 x 1012 | 36 | 1:75 |
| Streptavidin | 15nm | 4.0 | 20 | 5 x 1012 | 45 | 1:75 |
| Poly-L-Lysine | 15nm | 4.0 | 60 | 5 x 1012 | unknown | 1:75 |
| Immunoglobulin | 20nm | 4.0 | 30 | 2 x 1012 | 48 | 1:50 |
| Protein A | 20nm | 4.0 | 10 | 2 x 1012 | 64 | 1:50 |
| Streptavidin | 20nm | 4.0 | 20 | 2 x 1012 | 80 | 1:50 |
| Poly-L-Lysine | 20nm | 4.0 | 50 | 2 x 1012 | unknown | 1:50 |
| Immunoglobulin | 30nm | 5.0 | 15 | 8 x 1011 | 86 | 1:25 |
| Immunoglobulin | 40nm | 5.0 | 10 | 4.5 x 1011 | 150 | 1:25 |
| Notes: Gold conjugates 5nm to 30nm are packaged with 20% glycerol and can be frozen. 1nm conjugates are not packaged with glycerol and should not be frozen. EM Gold conjugates - %CV: <12% for 5nm; <8% for 10,15 & 20nm; <20% for 30nm . %Singlets: >85% for all conjugates except Protein A >95%. |
These gold conjugates are made to the highest standards and specifications, yielding excellent results when correctly used. The guidelines are straightforward and intended for use with either light or electron microscopy immunolabeling applications. All gold conjugates are supplied in one of the two buffers listed below. Buffer constituents dictate the shelf life and storage conditions for the individual conjugates.
Electron microscopy (EM) conjugates are supplied in the following buffer: 20mM Tris (tris-hydroxymethyl-aminomethane); 20mM sodium azide; 154mM NaCl; 20% glycerol; pH 8.2. Recipe to make 100ml: 0.242g (20mM) Tris + 0.9g (154mM) NaCl + ultrapure water to make 100ml. Adjust pH from 7.2 to 8.2 with 1N HCl or 1N NaOH. Storage: Stable for 1 year at 4°C; stability for 2+ years at –20°C. The conjugates demonstrate remarkable stability at ambient temperatures for up to 7 days. Repeat freezing and thawing is not recommended.
Ultra small (2nm) gold conjugates are supplied in the following buffer: 10mM Na2HPO4; 3mM KH2PO4; 123mM NaCl; 1% glycerol; 20mM sodium azide; pH 7.2. Recipe to make 100ml: 0.148g Na2HPO4 + 0.042g KH2PO4 + 0.72g NaCl + ultrapure water to make 100ml. Adjust pH from 7.2 to 8.2 with 1N HCl or 1N NaOH. Storage: Stable for 1 year at 4°C - DO NOT FREEZE. The conjugates demonstrate remarkable stability at ambient temperatures for up to 7 days.
Product Information
Each gold conjugate has a technical data sheet which indicates the following information: 1) Number of particles counted; 2) Mean particle diameter; 3) Coefficient of variation given as a percent; 4) Percent of single particles; 5) Percent of particles larger than triplets; and 6) Minimum detectable protein. The coefficient of variation is an important parameter in describing the relative distribution of gold particle sizes around the mean for a given batch. The coefficient of variation equals the standard deviation divided by the mean.
Normal Gaussian distributions work as follows: ±1 standard deviation describes 68% of the area under the curve; ± 2 standard deviations describe 95% of the area under the curve; ± 3 standard deviations describe 99.73% of the area under the curve. As an example, you have purchased a gold conjugate - Goat anti-Rabbit IgG (H+L), 10nm - having a mean particle diameter of 9.8nm with a coefficient of variation of 4.1%. First, the standard deviation needs to be determined. In this case it is 0.402nm (4.1% x mean particle diameter). Statistically, 68% of the particles will be from 9.40 to 10.20nm, 95% from 9.00 to 10.60nm and 99.73% from 8.60 to 11.00nm. A reliable size characterization has been determined for the batch.
Sample Fixation
All antigens and tissue react differently to different fixatives and preservation strategies (e.g. high pressure freezing, cryoultramicrotomy, etc.). As a result no one method will produce results all the time. Fixation is a very important step in determining the success of most immunolabeling procedures. Paraformaldehyde-based fixatives are most often used in concentrations from 0.5% up to 4% or higher. Often glutaraldehyde is added in concentrations typically from 0.1 to 0.5%. Cacodylate and the phosphate (Millonig, Sörensen) buffers are popular and the zwitterionic buffers such as HEPES or PIPES can be used as well for aldeyde fixation. The quenching of unreacted aldehydes is recommended by one of the following methods: 1) 0.05-0.1M ammonium chloride in buffer at 4°C for a few hours; 2) 0.1-0.2M glycine in buffer for a few hours; or 3) Sodium borohydride (0.1 mg/ml) in buffer for a few hours. Osmium tetroxide is not often used for tissues processed for immunolabeling. Berryman and Rodewald, 1990, J. Histochem, Cytochem., 38: 159-170 describe an excellent method to attain membrane contrast without the use of osmium.
Resin Infiltration/Polymerization
The hydrophilic polar resins (Lowicryl K4M and K11M; L R White; L R Gold) have proven to be the best based on immunoreactivity. Cryoultramicrotomy methods are more difficult but yield excellent results when done correctly. The polar resins are partially water soluble and do not need to be dehydrated to 100%. Apolar hydrophobic resins (Lowicryl HM20 and HM23) are available for use as well. The epoxy resins, for most applications, are not recommended. Dehydration, infiltration and polymerization with polar and apolar resins can be done at room temperatures or in the cold (progressively lower temperature techniques). Polymerization of polar and apolar resins is done by UV for immuno applications; however, UV as well as oven cures can be done with LR White.
Positive/Negative Controls
Positive as well as negative controls should be a part of all immunolabeling studies. Reagent and system dynamics require that controls be used to identify optimum signal-to-noise ratios (i.e. correct dilution of primary and secondary antibodies), viability of antibodies being used and necessary blocking steps or strategies. Tween 20® (Product 15713), Fish gelatin (45%) (Product15717) are good generic blocking reagents that can be mixed with antibody or rinse solutions.
Sample Labeling Protocol
Immunolabeling should be done in a humid chamber to protect against drying. The steps are:
1. Blocking step: 20-30 minutes at room temperature (recommend 0.1% Tween 20® and 0.1% Fish Gelatin to start. Add BSA 1-5% and the appropriate normal serum to reduce background if present).
2. Primary Antibody Incubation: 30-60 minutes at room temperature or 37°C.
3. Buffer Rinse: 30 minutes at room temperature with a buffer change every 5 minutes.
4. Gold Conjugate Incubation: 30-45 minutes in duration at room temperature or 37°C.
5. Buffer Rinse: A method found beneficial to reduce background is to elevate the NaCl concentration to approximately 2.5M from 1.54mM. The change in molarity has a beneficial effect on background due to ionic interaction of the gold conjugate (net negative change) and the substrate. Rinse for approximately 10 minutes and switch to water (ultrapure) for at least four changes of five minutes each.
6. Post Stain: 2% uranyl acetate (aqueous or alcoholic) for 5 minutes, rinse well in ultrapure water and then stain for 5 minutes with lead (25ml DI water, dissolve one sodium hydroxide pellet and add 0.125g of lead citrate). Rinse well after lead staining.
NOTE: Elevated temperatures (i.e. 37°C) during incubations require less time. Less time helps to keep the sections on the grids. Nickel or gold grids should be used. All grids, regardless, should be cleaned prior to use (1N HCl; 100% ETOH; 100% Acetone - Dry in 60°C oven on filter paper).
Questions and Customer Support: If there are any questions, please contact Ted Pella, Inc. and ask for Cindy Smith, Technical Product Specialist.